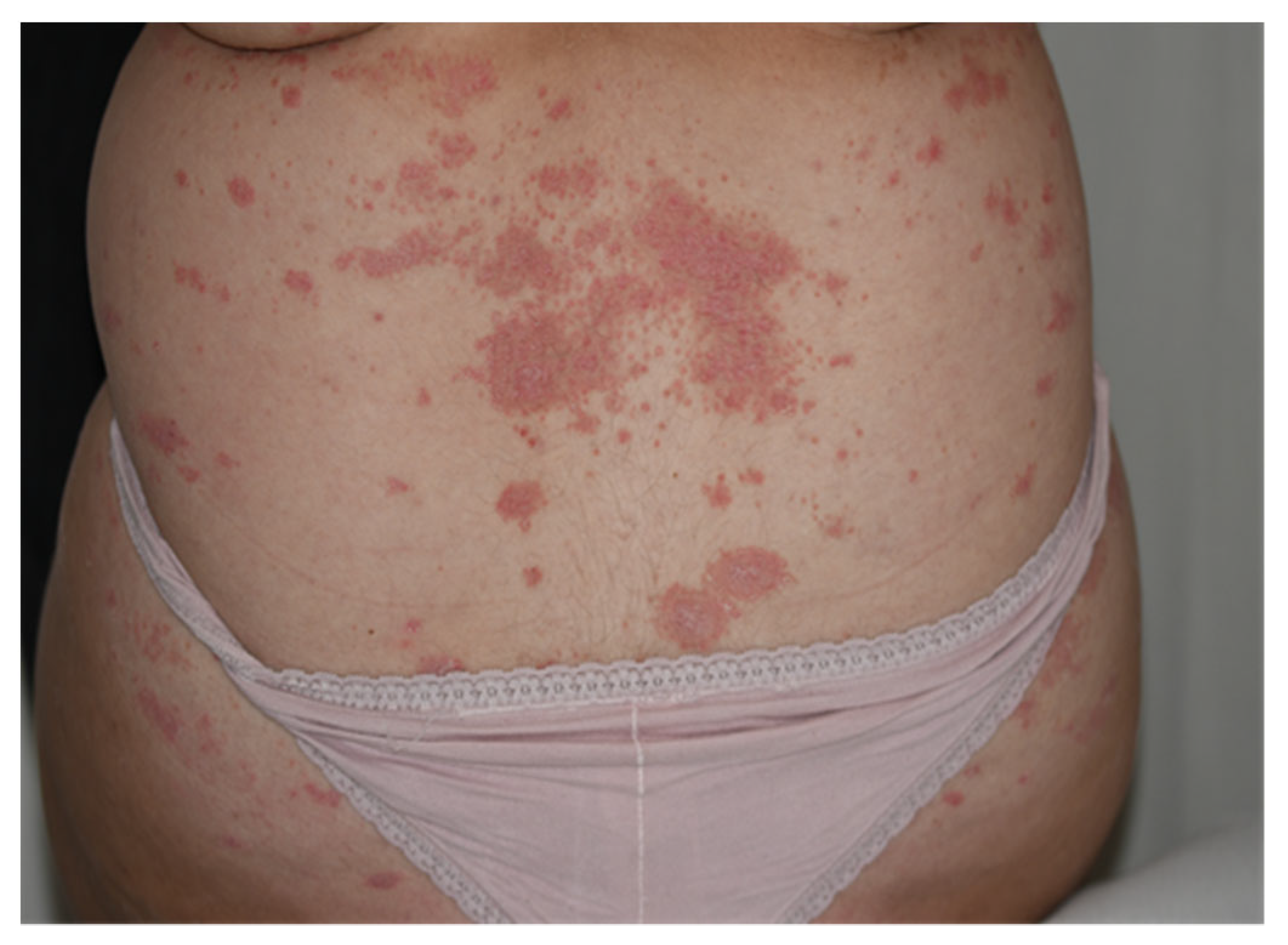
These highlights do not include all the information needed to use STELARA® safely and effectively. See full prescribing information for STELARA®. STELARA® (ustekinumab) injection, for subcutaneous or intravenous use Initial U.S. Approval:

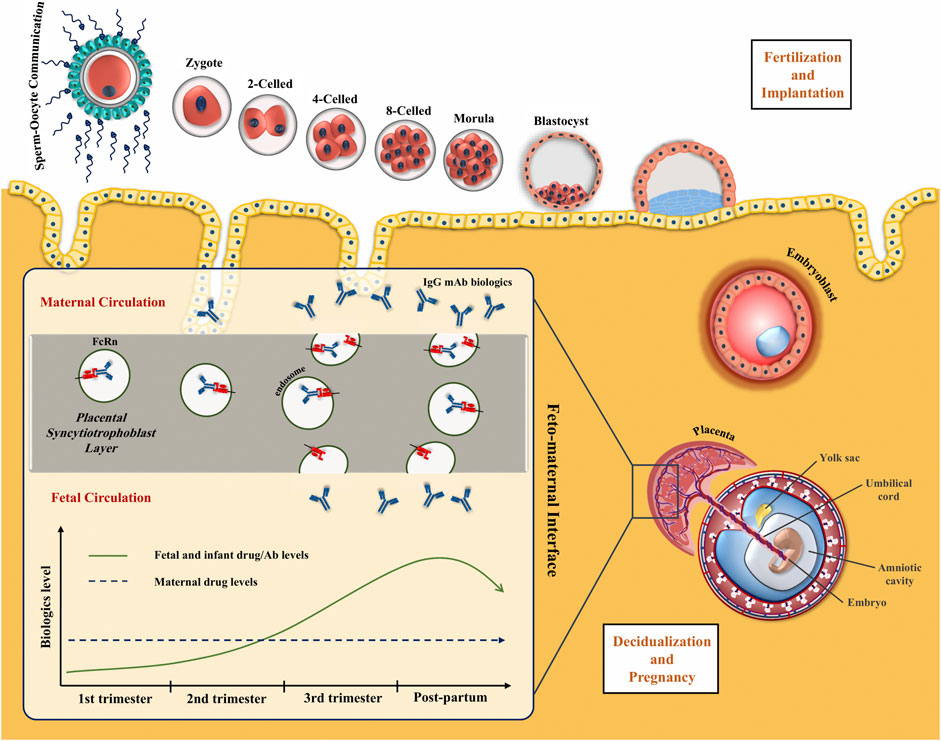
Infant Ustekinumab Clearance, Risk of Infection, and Development After Exposure During Pregnancy - Clinical Gastroenterology and Hepatology

Ustekinumab versus adalimumab for induction and maintenance therapy in biologic-naive patients with moderately to severely active Crohn's disease: a multicentre, randomised, double-blind, parallel-group, phase 3b trial - The Lancet